Expert Corner
Autotransplantation of mesenchymal stem cells in femoral head osteonecrosis
 Alexander E. Murzich
Alexander E. MurzichCo-authors: H.A.Zhernasechanka, M.A. Gerasimenko
Introduction
Surgical treatment of femoral head osteonecrosis is preferable at the early stages in order to preserve the structure and function of the hip joint especially in young patients aged 25–45 years. A reduction in the activity of osteoblasts and mesenchymal stem cells (MSCs), which are considered precursors of osteoblasts in the femoral head have been associated with the pathogenesis of osteonecrosis [1]. In fact, there is complete absence of MSCs in the necrotic lesion itself and significant reduction in the number of cells in the proximal part of the femur adjacent to the area of necrosis [1].
The use of autologous MSCs is a new and promising area in orthopedic surgery and has several advantages [2]. MSCs not only differentiate into osteoblasts in vivo, but also induce proliferation and differentiation of osteocyte precursor cells in the bone tissue that surrounds the necrotic region, thus stimulating the regeneration of the necrotic area [3]. Bone marrow MSCs have several advantages: the provision of a source for MSCs, the high proliferative potential and the high plasticity of cells with a tendency to differentiate in the osteogenic direction due to lack of immunogenicity, homing of MSCs to the site of injury and the paracrine effect of MSCs [4].
We developed a new method of autotransplantation of the MSCs for the surgical restoration of bone tissue in avascular necrosis of the femoral head in adults.
Preparation
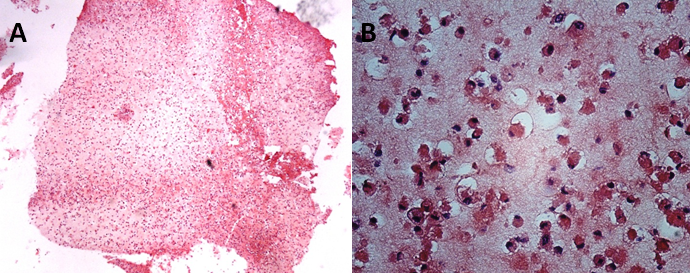
At the first stage, a protocol for osteogenic differentiation of bone marrow MSCs has been developed. We performed the experimental studies on animals. Dog bone marrow was utilised to obtain MSCs. Five biocomposites in vitro were obtained containing on average 19.0 x 106 osteogenically pre-differentiated cells. The biocomposites were transplanted into a pre-executed canine femoral head defect for bone repair and served as a control group. The histological section of the MSCs biocomposite in the fibrin gel was characterised by the presence of an eosinophilic amorphous substance with a large number of monomorphic cells, small eosinophilic rounded cytoplasm and an eccentric hyperchromic nucleus. Populated cells were evenly distributed in the thickness of the carrier (Figure 1).
At the second stage, eight patients with femoral head osteonecrosis were operated on using MSCs. According to the ARCO classification, in 6 patients the stage of the disease was II (sclerotic, cystic or osteoporotic changes), in two – IIIa (crescent sign, no collapse). Bone marrow was taken from the iliac crest in an average volume of 64.4 ± 4.3 mls. The cells product was manufactured for 4 weeks in the laboratory. An average of 32.1 ± 3.4 x 106 MSCs were obtained, colonised on a fibrin gel and then differentiated into osteocytes.
Surgical technique (Figure 2)
Under X-ray control, an Ilizarov guide wire is inserted through the femoral neck to the focus of necrosis. A bone graft is then taken from the intertrochanteric femoral region using hollow cutters with a diameter of 10-12 mm. Next, the affected area of the femoral head is drilled with cutters and tunneled with a spoke from the inside of the bone. To control the quality of decompression and for visual assessment of bone viability, an arthroscope is inserted into the femoral neck. Necrotic bone fragments are removed using a shaver under direct vision. A composite of MSCs in a fibrin gel is then mixed with the cancellous bone graft and introduced into the focus of necrosis percutaneously with the piston-like pusher. Then bone graft is inserted into the outside of the femoral neck canal.
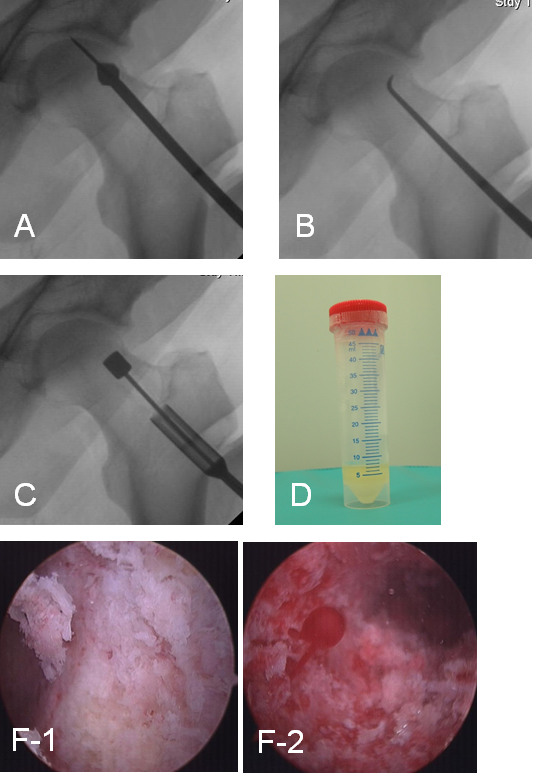
A– necrotic zone drilling, B– tunneling by spoke, C– composite of MSCs injection, D– MSCs product, F– arthroscopic view of the necrotic zone before (F-1) and after tunneling (F-2) showing intraosseous bleeding.
Results
The histological study of the microslides from the canine femoral heads showed the formation of mature bone tissue with osteoblasts at the site of MSCs injection.
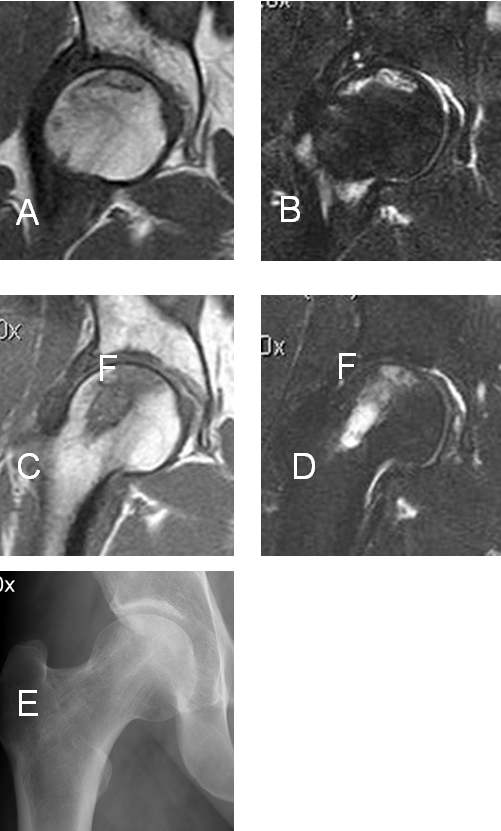
Our method for producing the MSCs biotransplant in fibrin gel allows us to save up to 94% of the cells in the composite at the time of transplantation to the patient. The clinical, radiological and MRI results were studied within one year. The mean Harris score before surgery was 73 ± 3.1 and in the postoperative period 84.2 ± 2.2. In one case, progression of collapse was observed. We didn’t observe any complications after MSCs transplantation. In 4 patients (50% of cases), a decrease of the bone marrow femoral head oedema 6-12 months after decompression was detected on MRI, consequently they had decrease in pain. In two cases (25%), a decrease in the severity of synovitis of the hip joint after surgery was observed. A clinical example is shown in Figure 3.
Conclusions
The osteoinductive effect of MSCs promotes bone tissue regeneration in the area of the affected femoral head. This method can be used in young 25-44 years patients with femoral head osteonecrosis and ARCO stages I-II, and IIIa when bacterial and infectious causes of the disease are excluded and there is vascular aetiology of osteonecrosis. We believe that autotransplantation of the MSCs during decompression makes it possible to improve the results of treatment of femoral head osteonecrosis and delay THA for years.
- Homma Y, Kaneko K, Hernigou P. Supercharging allografts with mesenchymal stem cells in the operating room during hip revision. International Orthopaedics. 2013;38(10):2033-2044
- Hernigou P, Flouzat-Lachaniette CH, Delambre J et al. Osteonecrosis repair with bone marrow cell therapies: state of the clinical art. Bone. 2015;70:102–109.
- Lebouvier A, Poignard A, Cavet M et al. Development of a simple procedure for the treatment of femoral head osteonecrosis with intra-osseous injection of bone marrow mesenchymal stromal cells: study of their biodistribution in the early time points after injection. Stem Cell Research & Therapy. 2015;6:68
- Gangji V, Maertelaer V De, Hauzeur J P. Autologous bone marrow cell implantation in the treatment of non-traumatic osteonecrosis of the femoral head: Five year follow-up of a prospective controlled study. Bone. 2011; 49:1005–1009.


















